Research
Prenatal delta-9-tetrahydrocannabinol exposure alters fetal neurodevelopment in rhesus macaques
Prenatal cannabis use is associated with adverse offspring neurodevelopmental outcomes; however the underlying mechanisms are relatively unknown. We sought to determine the impact of chronic delta-9-tetrahydrocannabinol (THC) exposure on fetal neurodevelopment in a rhesus macaque model using advanced imaging combined with molecular and tissue studies. Animals were divided into two groups, control (n = 5) and THC-exposed (n = 5), which received a daily THC edible pre-conception and throughout pregnancy. Fetal T2-weighted MRI was performed at gestational days 85 (G85), G110, G135 and G155 to assess volumetric brain development. At G155, animals underwent cesarean delivery with collection of fetal cerebrospinal fluid (CSF) for microRNA (miRNA) studies and fetal tissue for histologic analysis. THC exposure was associated with significant age by sex interactions in brain growth, and differences in fetal brain histology suggestive of brain dysregulation. Two extracellular vesicle associated-miRNAs were identified in THC-exposed fetal CSF; pathway analysis suggests that these miRNAs are associated with dysregulated axonal guidance and netrin signaling. This data is indicative of subtle molecular changes consistent with the observed histological data, suggesting a potential role for fetal miRNA regulation by THC. Further studies are needed to determine whether these adverse findings correlate with long-term offspring neurodevelopmental health.
Read more of publication, Prenatal delta-9-tetrahydrocannabinol exposure alters fetal neurodevelopment in rhesus macaques, on PubMed.
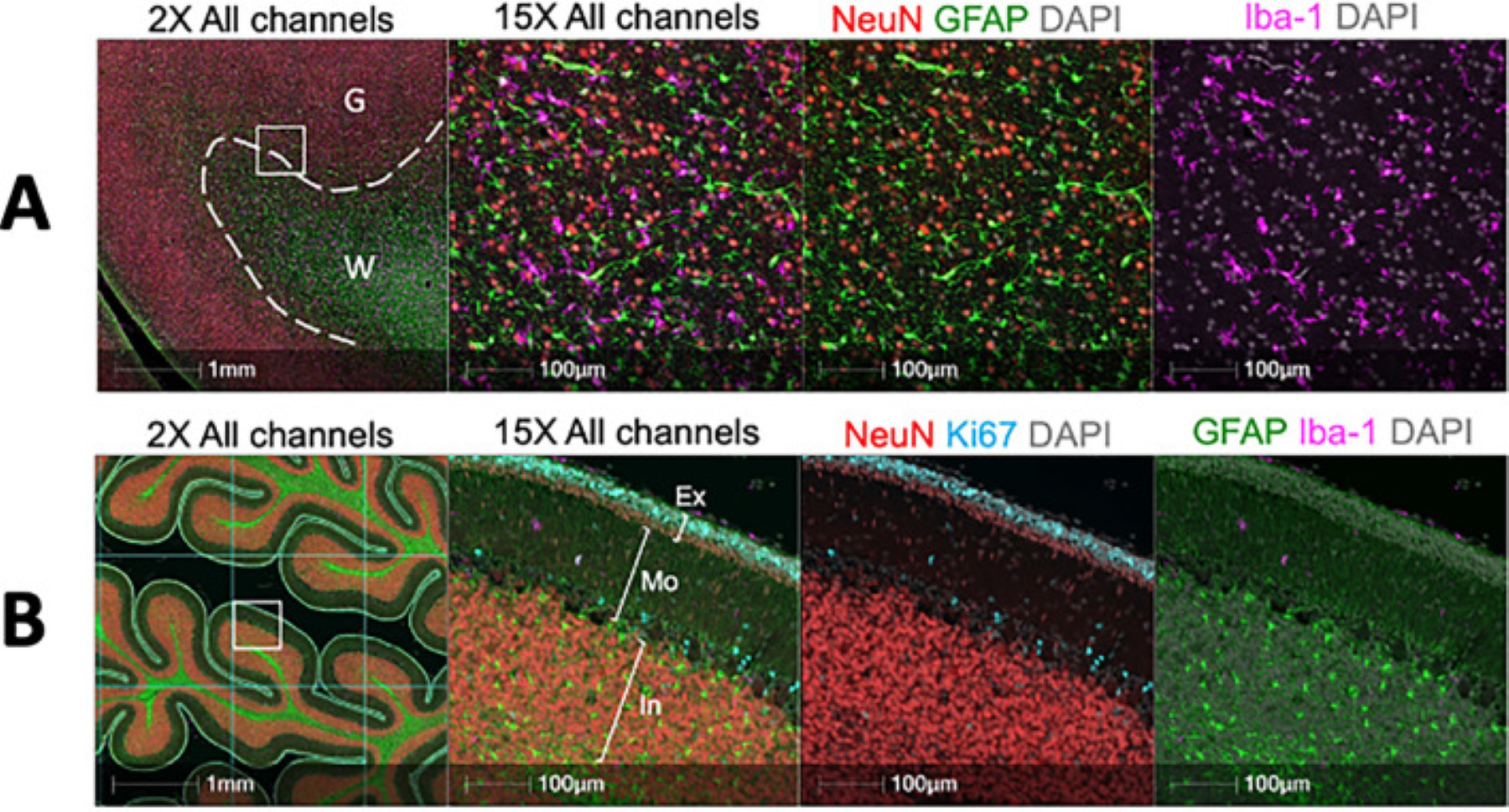
B Muliplex immunofluorescence image of the fetal cerebellum. FFPE sections were stained for neurons (NeuN—Red). Microglia (Iba-1—Magenta), Ki67 (Cyan) and astrocytes (GFAP—Green) by multiplex immunofluorescence. The external granule layer (Ex), Molecular layer (Mo) and internal granule layer (In) are highlighted in the 15× panel. For quantification, the tissue was divided by a grid and annotations representations representing each layer were drawn in 10 randomly selected grid squares per cerebellum.
Prenatal THC exposure is associated with changes in rhesus macaque DNA methylation enriched for autism genes
Background: With the growing availability of cannabis and the popularization of additional routes of cannabis use beyond smoking, including edibles, the prevalence of cannabis use in pregnancy is rapidly increasing. However, the potential effects of prenatal cannabis use on fetal developmental programming remain unknown.
Results: We designed this study to determine whether the use of edible cannabis during pregnancy is deleterious to the fetal and placental epigenome. Pregnant rhesus macaques consumed a daily edible containing either delta-9-tetrahydrocannabinol (THC) (2.5 mg/7 kg/day) or placebo. DNA methylation was measured in 5 tissues collected at cesarean delivery (placenta, lung, cerebellum, prefrontal cortex, and right ventricle of the heart) using the Illumina MethylationEPIC platform and filtering for probes previously validated in rhesus macaque. In utero exposure to THC was associated with differential methylation at 581 CpGs, with 573 (98%) identified in placenta. Loci differentially methylated with THC were enriched for candidate autism spectrum disorder (ASD) genes from the Simons Foundation Autism Research Initiative (SFARI) database in all tissues. The placenta demonstrated greatest SFARI gene enrichment, including genes differentially methylated in placentas from a prospective ASD study.
Conclusions: Overall, our findings reveal that prenatal THC exposure alters placental and fetal DNA methylation at genes involved in neurobehavioral development that may influence longer-term offspring outcomes. The data from this study add to the limited existing literature to help guide patient counseling and public health polices focused on prenatal cannabis use in the future.
Read more of the publication, Prenatal delta-9-tetrahydrocannabinol exposure is associated with changes in rhesus macaque DNA methylation enriched for autism genes, on Pubmed.
Cessation of chronic THC exposure partially reverses impacts on male fertility and the sperm epigenome in rhesus macaques
Chronic THC use resulted in significant testicular atrophy, increased gonadotropin levels, decreased serum sex steroid levels, changes in seminal fluid proteome, and increased DNA fragmentation with partial recovery after discontinuation of THC use. For every increase of 1 mg/7 kg/day in THC dosing, there was a significant decrease in the total testicular volume bilaterally by 12.6 cm3 (95% confidence interval [CI], 10.6-14.5), resulting in a 59% decrease in volume. With THC abstinence, the total testicular volume increased to 73% of its original volume. Similarly, with THC exposure, there were significant decreases in the mean total testosterone and estradiol levels and a significant increase in the follicle-stimulating hormone level. With increasing THC dose, there was a significant decrease in the liquid semen ejaculate volume and weight of coagulum; however, no other significant changes in the other semen parameters were noted. After discontinuing THC use, there was a significant increase in the total serum testosterone level by 1.3 ng/mL (95% CI, 0.1-2.4) and estradiol level by 2.9 pg/mL (95% CI, 0.4-5.4), and the follicle-stimulating hormone level significantly decreased by 0.06 ng/mL (95% CI, 0.01-0.11). Seminal fluid proteome analysis revealed differential expression of proteins enriched for processes related to cellular secretion, immune response, and fibrinolysis. Whole genome bisulfite sequencing identified 23,558 CpGs differentially methylated in heavy-THC vs. pre-THC sperm, with partial restoration of methylation after discontinuation of THC use. Genes associated with altered differentially methylated regions were enriched for those involved in the development and function of the nervous system.
Conclusion(s): This is the first study demonstrating that discontinuation of chronic THC use in rhesus macaques partially restores adverse impacts to male reproductive health, THC-associated sperm differentially methylated regions in genes important for development, and expression of proteins important for male fertility.
Read more of the publication, Cessation of chronic delta-9-tetrahydrocannabinol use partially reverses impacts on male fertility and the sperm epigenome in rhesus macaques, on PubMed
Chronic prenatal THC exposure adversely impacts placental function and development in a rhesus macaque model
Cannabis use in pregnancy is associated with adverse perinatal outcomes, which are likely mediated by the placenta. However, the underlying mechanisms and specific vasoactive effects of cannabis on the placenta are unknown. Our objective was to determine the impact of chronic prenatal delta-tetrahydrocannabinol (THC, main psychoactive component of cannabis) exposure on placental function and development in a rhesus macaque model using advanced imaging. Animals were divided into two groups, control (CON, n = 5) and THC-exposed (THC, n = 5). THC-exposed animals received a THC edible daily pre-conception and throughout pregnancy. Animals underwent serial ultrasound and MRI at gestational days 85 (G85), G110, G135 and G155 (full term is ~ G168). Animals underwent cesarean delivery and placental collection at G155 for histologic and RNA-Seq analysis. THC-exposed pregnancies had significantly decreased amniotic fluid volume (p < 0.001), placental perfusion (p < 0.05), and fetal oxygen availability (p < 0.05), all indicators of placental insufficiency. Placental histological analysis demonstrated evidence of ischemic injury with microinfarctions present in THC-exposed animals only. Bulk RNA-seq demonstrated that THC alters the placental transcriptome and pathway analysis suggests dysregulated vasculature development and angiogenesis pathways. The longer-term consequences of these adverse placental findings are unknown, but they suggest that use of THC during pregnancy may deleteriously impact offspring development.
Read more of this publication, Chronic prenatal delta-9-tetrahydrocannabinol exposure adversely impacts placental function and development in a rhesus macaque model, on PubMed.
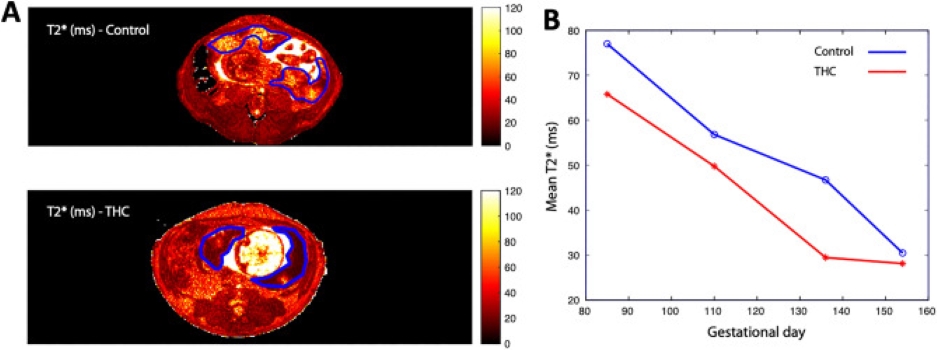
Histogram plot of T2* versus percent of placental voxels displayed for THC-exposed (red) vs. control animals (blue) at G85, G110, G135 and G155. (A) Quantitative T2* maps of placental blood flow of an axial MRI-image at the level of the uterus of a representative control (top) and THC-exposed (bottom) animal with the bi-lobed rhesus macaque placenta outlined in blue. (B) THC-exposed animals had a smaller fraction of large T2* values compared to controls across all time points, demonstrating decreased placental perfusion and fetal oxygen availability in the former. *p = 0.04 for THC effect by repeated measures ANOVA.
Chronic delta-9-tetrahydrocannabinol exposure impacts testicular volume and male reproductive health in rhesus macaques
For each 1 mg/7 kg/day increase in THC dosing, there was a marked loss in total bilateral testicular volume of 11.8 cm3 (95% confidence interval [CI]: 8.3-15.4). In total, average bilateral testicular volume decreased by 58%. Significant dose-response decreases in mean total testosterone level by 1.49 ng/mL (95% CI: 0.83-2.15) and in estradiol level by 3.8 pg/mL (95% CI: 2.2-5.4) were observed, but significant increases in the levels of follicle-stimulating hormone by 0.06 ng/mL (95% CI: 0.02-0.10), luteinizing hormone by 0.16 ng/mL (95% CI: 0.08-0.25), and prolactin by 7.4 ng/mL (95% CI: 3.4-11.3) were observed. There were no statistically significant changes in semen parameters.
Conclusion(s): In rhesus macaques, chronic exposure to THC resulted in significant dose-response testicular atrophy, increased serum gonadotropin levels, and decreased serum sex steroids, suggestive of primary testicular failure. Further studies are needed to determine if reversal of these observed adverse effects would occur if THC was discontinued and for validation of the findings in a human cohort.
Read more of this publication, Chronic exposure to delta-9-tetrahydrocannabinol impacts testicular volume and male reproductive health in rhesus macaques, on PubMed.